Відділ Біофізики сенсорної сигналізації
(→Генетична корекція больової чутливості у щурів з хронічним болем) |
(→Members) |
||
| Line 155: | Line 155: | ||
|bagatskaya@biph.kiev.ua | |bagatskaya@biph.kiev.ua | ||
|380442562082 | |380442562082 | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|12. | ||
| + | |д.б.н. Півнева Т.А. | ||
| + | | | ||
| + | | | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|13. | ||
| + | |Романенко С.В. | ||
| + | | | ||
| + | | | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|14. | ||
| + | |Дужий Д.Є | ||
| + | | | ||
| + | | | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|15. | ||
| + | |Костюк О.І. | ||
| + | | | ||
| + | | | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|16. | ||
| + | |Яценко В.П. | ||
| + | | | ||
| + | | | ||
| + | |-style="background:#ccffcc;" | ||
| + | |style="text-align:left;"|17. | ||
| + | |Мітрузаєва О.А. | ||
| + | | | ||
| + | | | ||
|-style="background:#ccffcc;" | |-style="background:#ccffcc;" | ||
|style="text-align:left;"| | |style="text-align:left;"| | ||
|} | |} | ||
Revision as of 13:32, 25 October 2023
Загальна інформація
Головними предметами досліджень протягом останніх років були механізми регуляції внутрішньоклітинної концентрації Ca2+та кальцієва сигналізації в різних типах збудливих клітин.В експериментах використовуються нейрони, які є ізольованими або знаходяться в культурі, а також зрізи мозкута інтактний спинний мозок.В наших дослідженнях ми застосовуємо наступні експериментальні методи: оптичні (цифрова та конфокальна мікроскопія), електрофізіологічні (мікроелектроди, іонофорез, петч-клемп), генетичні (трансфекція, інфекція, сайт-специфічний мутагенез) та математичні(математичне моделювання, математична статистика).
Дослідження
Signalling of neuronal Ca2+ sensor proteins
Decoding glutamate receptor activation by the Ca2+ sensor proteins
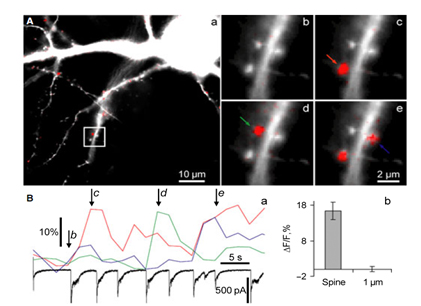
We study biophysical and physiological mechanisms of NCS (Neuronal Calcium Sensor) protein signaling in hippocampal neurons. Decoding of complex spatio-temporal patterns of [Ca2+]i changes by a Ca2+ sensor proteins, hippocalcin and neurocalcin δ, and involvement of Ca2+-dependent protein translocation in neuronal signal transduction is in a center of these studies. The most important achievement in this line of research is clear understanding that hippocalcin and neurocalcin δ may differentially decode various spatiotemporal patterns of glutamate receptor activation into site- and time-specific translocation to their membranous targets. Hippocalcin also possesses an ability to produce local signalling at the single synaptic level providing a molecular mechanism for homosynaptic plasticity (Dovgan et al., 2010). Examples of spontaneous hippocalcin-YFP translocation to a set of spines correlated with bursts of postsynaptic currents are shown in the Figure 1. У відділі також досліджується функціонування кальційрегулюючих систем, зокрема кальцієвих каналів та глутаматних рецепторів, в ноцицептивних нейронах задньокорінцевих гангліїв та заднього рогу спинного мозку в різних моделях нейропатичного та запального болю. Розуміння функціонування цих механізмів важливо для розробки новітніх підходів у лікуванні болю різного генезу.
Analysis of the mechanisms underlying GABAergic synaptic transmission
Short-term plasticity of GABAergic synaptic transmission in the hippocampus
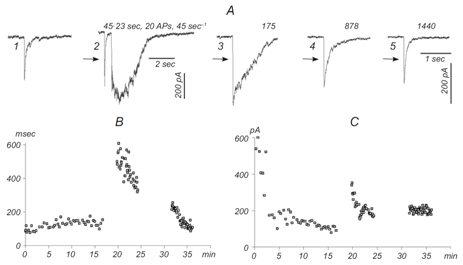
This part of our research is devoted to studies of different synaptic mechanisms contributing to GABAergic synaptic transmission in the hippocampus. Mechanisms of short-term depression and non-phasic vesicular release are in focus of our current research (Stepanyuk et al., 2014b)(Borisyuk et al., 2014). We have recently found a new type of short-term plasticity in cultured hippocampal neurons manifested as activity-dependent potentiation of an asynchronous component of GABAergic synaptic currents, аIPSCs (Fig. 2) (Borisyuk et al., 2014).
Simultaneously with potentiation of аIPSC, we observed plateau-like inward currents in the presynaptic neuron. The charge transferred by this current correlated significantly with the decay time of evoked IPSC in the postsynaptic neuron (mean correlation coefficient 0.83±0.10) suggesting that the inward current mediates аIPSC potentiation. We hypothesize that the observed plasticity may endogenously regulate the efficacy of GABAergic synaptic transmission in the hippocampus. In the ongoing research we are studying pre- and postsynaptic mechanisms involved in this new type of inhibitory synaptic plasticity.
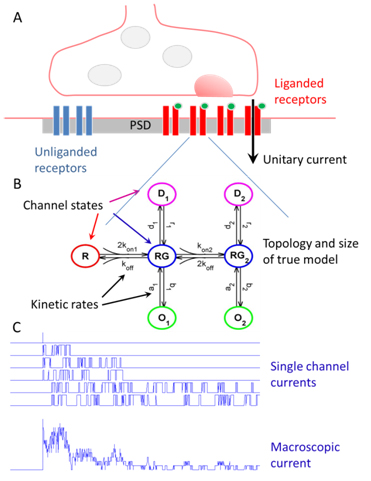
Maximum likelihood estimation of biophysical parameters of synaptic receptors from macroscopic currents
Dendritic integration and neuronal firing patterns strongly depend on biophysical properties of synaptic ligand-gated channels. However, precise estimation of biophysical parameters of these channels in their intrinsic environment is complicated and still unresolved problem. We have recently described novel methods based on a maximum likelihood approach that allows to estimate not only the unitary current of synaptic receptor channels but also their multiple conductance levels, kinetic constants, the number of receptors bound with a neurotransmitter, and the peak open probability from experimentally feasible number of postsynaptic currents (Stepanyuk et al., 2011)(Stepanyuk et al., 2014a). The new method also improves the accuracy of evaluation of unitary current as compared to the peak-scaled non-stationary fluctuation analysis, leading to a possibility to precisely estimate this important parameter from a few postsynaptic currents recorded in steady-state conditions. Estimation of unitary current with this method is robust even if postsynaptic currents are generated by receptors having different kinetic parameters, the case when peak-scaled non-stationary fluctuation analysis is not applicable. Thus, with the new method, routinely recorded postsynaptic currents could be used to study the properties of synaptic receptors in their native biochemical environment.
Further elaboration of this approach that we are doing now will lead to development of user-friendly software that may be widely used to quantitatively study interaction of synaptic channels with pharmacological agents and modulation of channel functioning in normal and pathological conditions.
Peripheral and central mechanisms of neuropathic pain
Role of T-type Ca2+ channels in central processes of nociceptive neurons in maintenance of diabetic neuropathy
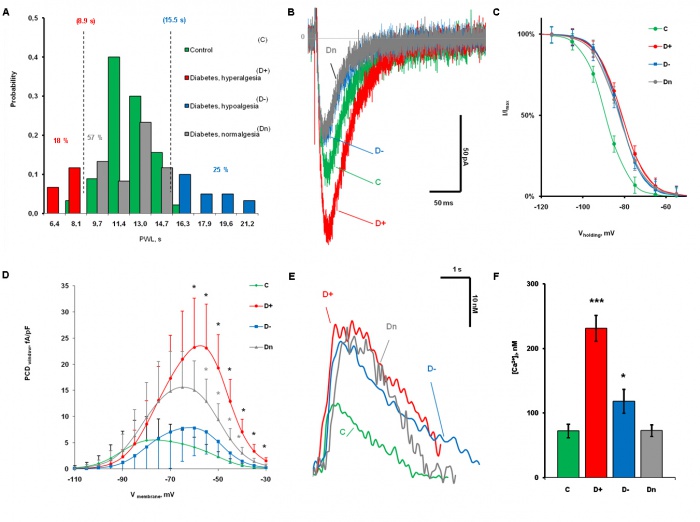
Role of T-type Ca2+ channels in central processes of nociceptive neurons in maintenance of diabetic neuropathy Peripheral diabetic neuropathy (PDN), occurring in about 60% - 70% of diabetic patients, is among the most severe consequences of diabetes. Chronic pain characterized by spontaneous deep aching or burning pain, as well as by mechanical allodynia and thermal hyperalgesia is one of the most disturbing symptoms of PDN. However, the specific cellular mechanisms responsible for development and maintenance of peripheral diabetic neuropathy are mainly unknown. We have recently found that different types of nociceptive DRG neurons demonstrate diabetes-induced upregulation of CaV3.2 subtype of T-type Ca2+ channels during different stages of diabetes development (Khomula et al., 2013)(Khomula et al., 2014)(Duzhyy et al., 2015). The upregulation of T-type channels resulted in the increased neuronal excitability of nociceptive neurons revealed by a lower threshold for action potential initiation, prominent afterdepolarizing potentials and burst firing. The upregulation T-type channels and increased excitability of neurons may contribute to thermal hyperalgesia in early diabetes and nonthermal nociception (e.g. mechanical allodynia and hyperalgesia and spontaneous pain) at its later-stage. We hypothesize that T-type Ca2+ channels are functionally expressed in central nociceptive processes and that upregulation of these channels under diabetic conditions amplifies peripheral action potentials in these neurons to busting discharges leading to changes in nociceptive output of lamina I projection neurons of the spinal cord and maintenance of painful diabetic neuropathy. Since direct electrophysiological measurements of T-type channel functioning in central axons of nociceptive DRG neurons are not methodologically feasible we are testing this hypothesis by an innovative combination of state-of-the-art approaches that include a novel whole spinal cord preparation that preserves the complex organization of dorsal roots in the spinal cord, Ca2+ imaging in identified central axons of specific types of primary nociceptive neurons using genetic Ca2+ indicators and profound computer modelling.
Peripheral and central mechanisms underlying inflammatory chronic pain
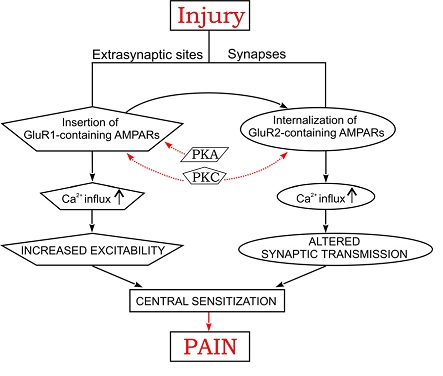
Previous research has resulted in general understanding of importance of Ca2+-dependent glutamate receptor (AMPA receptor subtype) trafficking in the dorsal horn of spinal cord as one of the possible mechanisms of inflammatory chronic pain (Kopach and Voitenko, 2013). It is summarized in Figure 4.
Continuing this research we have recently found that complete Freund adjuvant (CFA)-induced peripheral inflammation, a well-known model of chronic inflammatory pain, prominently augments excitatory neurotransmission in rat dorsal horn lamina II neurons exhibiting adapting firing patterns (Kopach et al., 2015) and apparently representing excitatory glutamatergic interneurons. At the same time this peripheral inflammation decreases excitatory drive to the tonic firing lamina II neurons most of which are inhibitory. The inhibitory drive is also increased to the inhibitory neurons and decreased to the excitatory ones as a result of the inflammation (Kopach et al., 2015). Thus, the balance between excitation and inhibition in the lamina II of dorsal horn is strongly shifted toward the excitation (Kopach et al., 2015). The lamina II interneurons directly synapse onto lamina I projection neurons (PNs), the main output of painful signaling in the spinal cord. Thus, these inflammatory-induced, neuron-type specific changes in synaptic activity in lamina II neurons most probably results in an increase of the excitatory drive and a decrease of the inhibitory drive to lamina I PNs. In its turn it may increase PN excitability, hence contributing to maintenance of the inflammatory pain. In this research we aim to test this hypothesis and to study what cellular and molecular mechanisms underlie increased excitability of PNs under inflammatory conditions.
Генетична корекція больової чутливості у щурів з хронічним болем
Діабет-індуковані зміни у функціонуванні кальцієвих каналів Т-типу в нейронах ДКГ
Recent References
- Agashkov K, Krotov V, Krasniakova M, Shevchuk D, Andrianov Y, Zabenko Y. Distinct mechanisms of signal processing by lamina I spino- parabrachial neurons. Sci Rep. 9(1):19231 (2019). PMID:31848358
- Osypenko, D. S. et al. Perturbed Ca2+-dependent signaling of DYT2 hippocalcin mutant as mechanism of autosomal recessive dystonia. Neurobiol. Dis. 132, 104529 (2019). PMID:31301343
- Krotov, V., Tokhtamysh, A., Safronov, B. V., Belan, P. & Voitenko, N. High-threshold primary afferent supply of spinal lamina X neurons. Pain 160, 1982–1988 (2019). PMID:30985620
- Gryshchenko O, Gerasimenko J V, Peng S, Gerasimenko O V, Petersen OH. Calcium signalling in the acinar environment of the exocrine pancreas: physiology and pathophysiology. J Physiol. 596(14):2663–78 (2018). PMID:29424931
- Bos R, Harris-Warrick RM, Brocard C, Demianenko LE, Manuel M, Zytnicki D, et al. Kv1.2 Channels Promote Nonlinear Spiking Motoneurons for Powering Up Locomotion. Cell Rep. 22(12):3315–27 (2018). PMID:29562186
- Cherkas, V. et al. Measurement of intracellular concentration of fluorescently-labeled targets in living cells. PLoS One 13, e0194031 (2018). PMID:29694385
- Delmas P, Korogod S, Coste B. Biophysical mechanisms of pathological activity of neurons related to hypoxia-ischemic and inflammatory injuries. In: Noxious Mechanosensation The Oxford Handbook of the Neurobiology of Pain. (2018). DOI:10.13989/j.cnki.0517-6611.2015.11.042
- Savchenko A, Cherkas V, Liu C, Braun GB, Kleschevnikov A, Miller YI, et al. Graphene biointerfaces for optical stimulation of cells. Sci Adv. 4(5):eaat0351 (2018). PMID:29795786
- Peng S, Gerasimenko J V, Tsugorka TM, Gryshchenko O, Samarasinghe S, Petersen OH, et al. Galactose protects against cell damage in mouse models of acute pancreatitis. J Clin Invest. 128(9):3769–78 (2018). PMID:29893744
- Krotov, V. et al. Functional Characterization of Lamina X Neurons in ex-Vivo Spinal Cord Preparation. Front. Cell. Neurosci. 11, 1–12 (2017). PMID:29163053
- Gryshchenko O, Gerasimenko J V, Gerasimenko O V, Petersen OH. Calcium signalling in pancreatic stellate cells: Mechanisms and potential roles. Cell Calcium 59(2–3):140–4 (2016). PMID:26960936
- Kopach, O. et al. HIF-1α-mediated upregulation of SERCA2b: The endogenous mechanism for alleviating the ischemia-induced intracellular Ca(2+) store dysfunction in CA1 and CA3 hippocampal neurons. Cell Calcium 59, 251–61 (2016). PMID:29163053
- Peng S, Gerasimenko J V, Tsugorka T, Gryshchenko O, Samarasinghe S, Petersen OH, et al. Calcium and adenosine triphosphate control of cellular pathology: asparaginase-induced pancreatitis elicited via protease-activated receptor 2. Philos Trans R Soc Lond B Biol Sci [Internet]. 371(1700):20150423 (2016). PMID:27377732
- Kopach, O., Krotov, V., Belan, P. & Voitenko, N. Inflammatory-induced changes in synaptic drive and postsynaptic AMPARs in lamina II dorsal horn neurons are cell-type specific. Pain 156, 428–38 (2015). PMID:25599231
- Duzhyy, D. E., Viatchenko-Karpinski, V. Y., Khomula, E. V, Voitenko, N. V & Belan, P. V. Upregulation of T-type Ca2+ channels in long-term diabetes determines increased excitability of a specific type of capsaicin-insensitive DRG neurons. Mol. Pain 11, 29 (2015). PMID:25986602
- Stepanyuk, A. R., Borisyuk, A. L., Tsugorka, T. M. & Belan, P. V. Different pools of postsynaptic GABAA receptors mediate inhibition evoked by low- and high-frequency presynaptic stimulation at hippocampal synapses. Synapse 68, 344–54 (2014). PMID:24677449
- Stepanyuk, A., Borisyuk, A. & Belan, P. Maximum likelihood estimation of biophysical parameters of synaptic receptors from macroscopic currents. Front. Cell. Neurosci. 8, 303 (2014). PMID:25324721
- Khomula, E. V, Viatchenko-Karpinski, V. Y., Borisyuk, A. L., Duzhyy, D. E., Belan, P. V & Voitenko, N. V. Specific functioning of Cav3.2 T-type calcium and TRPV1 channels under different types of STZ-diabetic neuropathy. Biochim. Biophys. Acta 1832, 636–49 (2013). PMID:23376589
- Kopach, O., Kao, S.-C. C., Petralia, R. S., Belan, P., Tao, Y.-X. X. & Voitenko, N. Inflammation alters trafficking of extrasynaptic AMPA receptors in tonically firing lamina II neurons of the rat spinal dorsal horn. Pain 152, 912–23 (2011). PMID:21282008
- Dovgan, a V, Cherkas, V. P., Stepanyuk, a R., Fitzgerald, D. J., Haynes, L. P., Tepikin, a V, Burgoyne, R. D. & Belan, P. V. Decoding glutamate receptor activation by the Ca2+ sensor protein hippocalcin in rat hippocampal neurons. Eur. J. Neurosci. 32, 347–58 (2010). PMID:20704590
Members
| Name | Work number | ||
|---|---|---|---|
| 1. | Belan P.V. | pasha@biph.kiev.ua | 380442562053 |
| 2. | Kononenko M.I. | ni.kononenko@biph.kiev.ua | 380442562428 |
| 3. | Gryshchenko O.V. | alesha@biph.kiev.ua | 380442562555 |
| 4. | Saftenku О.Е. | esaft@biph.kiev.ua | 380442562085 |
| 5. | Cherkas V.P. | cherkas@biph.kiev.ua | 380442562518 |
| 6. | Dovgan O.V. | rasboinik@mail.ru | 380442562428 |
| 7. | Borisyuk A.L. | borisyuk@biph.kiev.ua | 380442562518 |
| 8. | Krotov V.V. | vovakrotov@gmail.com | 380442562426 |
| 9. | Sheremet E. | sheremetik@yandex.ru | 380442562518 |
| 10. | Dromaretskiy A.V | avi9526@biph.kiev.ua | 380442562518 |
| 11. | Bagatska O.V. | bagatskaya@biph.kiev.ua | 380442562082 |
| 12. | д.б.н. Півнева Т.А. | ||
| 13. | Романенко С.В. | ||
| 14. | Дужий Д.Є | ||
| 15. | Костюк О.І. | ||
| 16. | Яценко В.П. | ||
| 17. | Мітрузаєва О.А. | ||